Automated Analysis Workstation
Automated microscopic analysis of fecal concentrates can be performed at reduced cost, higher accuracy and with enhanced worker safety by a new counter top clinical workstation. DiaSys Corporation of Waterbury, Connecticut has developed the FE-2, an easy-to-use workstation for rapid location and identification of parasites, cysts and ova in human feces.
Several years ago, information appearing in the pages of the NASA Tech Briefs publication assisted DiaSys's manufacturing of the R/S 2000, the company's first product. There is now a family of "R/S" workstations, all of which speed up, standardize, automate and make safer the laboratory analysis of urine sediment. The initial "R/S" workstation was made possible in part by coverage of work at the Jet Propulsion Laboratory and Langley Research Center. This stimulated a fast-paced roll-out of additional products from DiaSys.
The success of the R/S 2000 was followed by the R/S 1000, another product that standardizes automated urine sediment analysis for small hospital labs, physician group practices, "stat" labs, and outpatient clinics.
In September 1995, DiaSys introduced its third product, the R/S 2003. This larger, more advanced system standardizes and drives the cost out of routine urine sediment analysis conducted by larger labs and lab networks. The DiaSys workstations have been named the preferred practice by SmithKline Beecham Laboratories, Kaiser Permanente of Southern California, and many other hospitals and private lab groups.
Recently, a fourth workstation product was released to market--the FE-2. This latest technology is a workstation that automates the process of making and manipulating wet-mount preparations of fecal concentrates. A decrease in time needed to read the sample is achieved, permitting technologists to rapidly spot parasites, ova (eggs) and cysts, sometimes carried in the lower intestinal tract of humans and animals.
The FE-2 workstation corrects the procedural shortcomings of current methodologies in several significant ways. First of all, no special training is required to operate the FE-2. To operate, the technologist inserts the instrument's automatic dual aspirator into a prepared fecal concentrate and presses the "sample" button on the workstation's control console. Within 5 seconds, a consistent, measured amount of concentrate is automatically transferred to the workstation's optical slide assembly (OSA) on the stage of the microscope.
During this process, one chamber of the OSA is inoculated with a small quantity of the fecal concentrate. The other chamber is inoculated with an equal mix of fecal concentrate and iodine (for staining) or isotonic saline (for dilution) as the lab prefers. Observations of the stained (or diluted) and unstained fecal concentrate are then ready to begin. Purging the system takes about 7 seconds, after which time the FE-2 is ready to make the next concentrate slide.
Among its virtues, the FE-2 increases the level of safety and precision by automating the aspiration, resuspension, staining or diluting, transfer, presentation, and disposal of fecal concentrates. Eliminated is the need and cost of disposable pipettes, microscope slides, and cover slips. Moreover, because the fecal concentrate is contained in and manipulated through a sealed system, the technologist carrying out the work is not exposed to prolonged inhalation of chemicals such as formalin which are conventionally used to process raw fecal material.
Employing the FE-2 is non-invasive, can be performed on an out-patient basis, and quickly provides confirmatory results, says DiaSys Corporation president, Todd DeMatteo. "To the best of our knowledge, there is no competitive product or system in the market today," DeMatteo says.

DiaSys Corporation's FE-2 workstation eliminates the need and cost of disposable pipettes, microscope slides, and cover slips for faster and safer analysis of fecal concentrates.

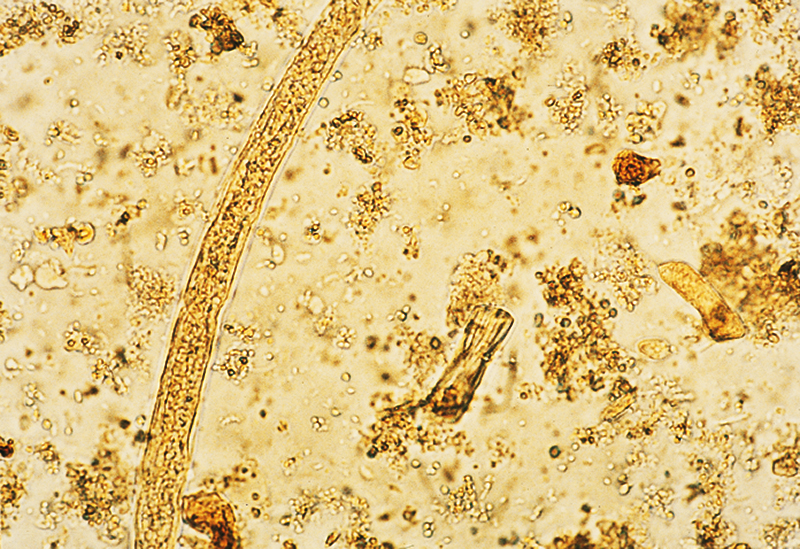
Parasites in a fecal sample (at a magnification of 100 power) can be detected by the FE -2. (Specimen courtesy of Dr. Dwight F. Miller at Saint Mary's Hospital in Waterbury, Connecticut.)