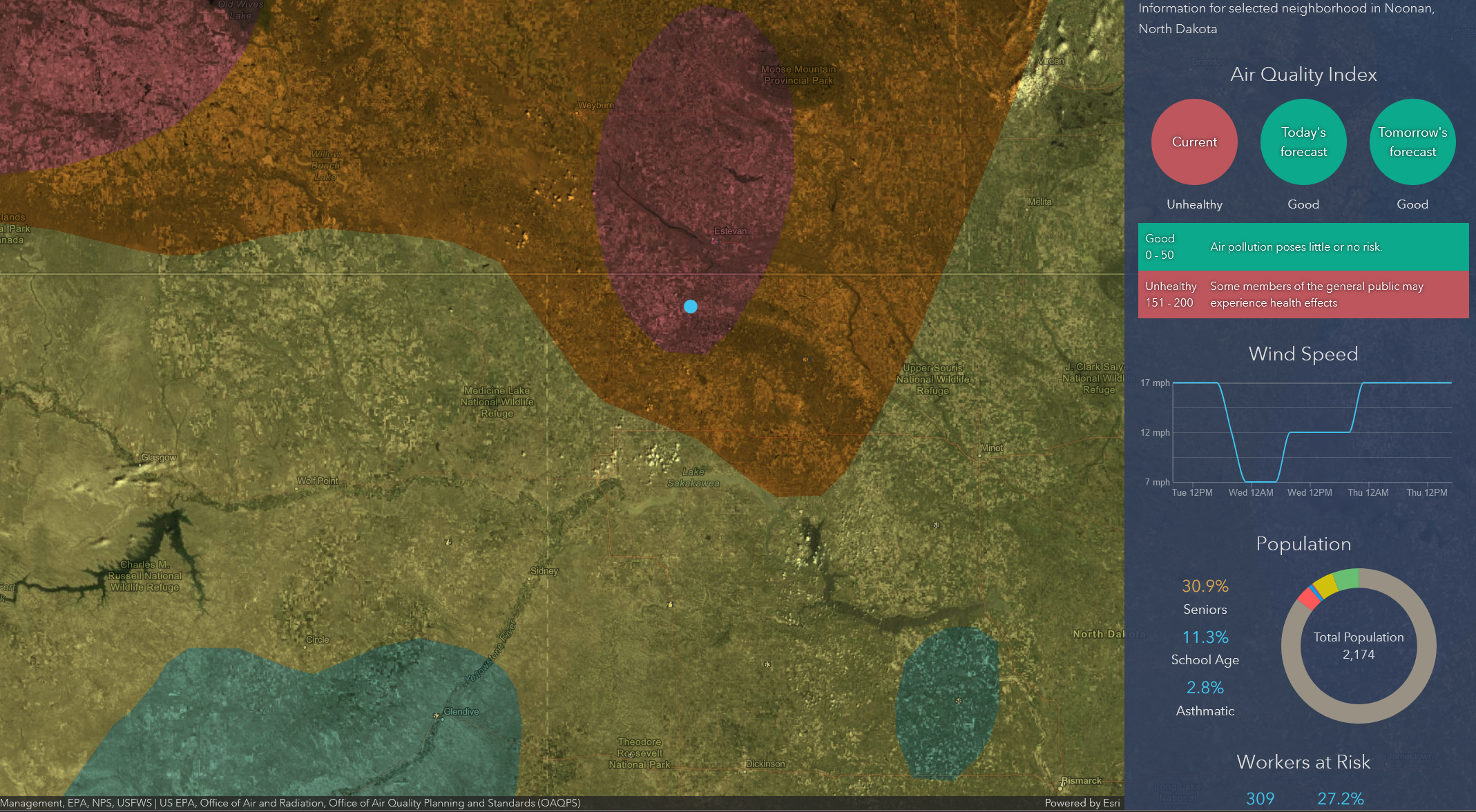
Versatile Fuel Cells Stop Natural Gas Emissions at Oil Wells
NASA Technology
In 1999, a NASA engineer and a professor from the California Institute of Technology (Caltech) ran into each other at a conference and hatched an idea. It would take two decades to bring to fruition, but the result is now stopping natural gas emissions at a handful of oil wells and is poised to spread throughout the industry.
Sekharipuram “Sri” Narayan had designed and patented a fuel cell that ran on methanol at NASA’s Jet Propulsion Laboratory (JPL), taking advantage of funds from the Department of Defense to come up with a power source that might also be useful for space missions. At the time, though, he was struggling with an issue with the cells: water in the electrolyte transports some of the methanol across the cell membrane that separates the electrodes, reducing efficiency.
Meanwhile, Sossina Haile, then a professor at Caltech in the Materials Science Department, had invented a solid acid crystal with a super power: at high temperatures it became, as she put it, “superprotonic,” meaning protons could pass through it at record speeds. Transferring protons happens to be an electrolyte’s job in a fuel cell, and this acid crystal not only could do that exceedingly well, but it also didn’t rely on the water that tends to misplace methanol in methanol fuel cells.
During a chance meeting at a conference in San Francisco, the two scientists realized their inventions might make a good combination. But this was the first Narayan had heard of solid acid crystals, and Haile’s team had no experience making or testing fuel cells. They agreed she would send two of her students to work with him. Calum Chisholm and Dane Boysen soon showed up at Narayan’s JPL laboratory with a quantity of the solid acid—cesium hydrogen sulfate was what they were working with at the time.
“I thought of a way to mix it with a polymer, hot press it, and create some free-standing membranes,” Narayan says. “It wasn’t very good, but it was functional.” Over the course of a few visits, he showed the pair of students how to apply electrodes at each end and create a structure to supply fuel. They were able to test it and show that it worked, even if the electrolyte was inclined to decompose under some conditions.
Today’s version uses a different solid acid electrolyte—cesium dihydrogen phosphate—but it’s still built using the same basic method Narayan taught the students 20 years ago.
Technology Transfer
Haile was excited by the results and started looking into other solid acids. She, Chisholm, and Boysen kept working on the cells, raised some money, and, in 2005, founded Superprotonic Inc. to build a commercial product. Narayan remained an occasional consultant. But four years and $14 million later, the recession forced them to shut down.
The Superprotonic years hadn’t been wasted, though. The team had scaled up their technology from a pile of nickel-sized cells with a lifetime of less than 100 hours to a stack of five-inch plates that could produce 50 watts of power for nearly 1,000 hours. Just months after shutting down, members of the former company secured a new development contract and founded SAFCell, based in Pasadena, California, with Chisholm as CEO.
The contract came from a Norwegian company interested in a 1.4-kilowatt fuel cell stack. The U.S. Department of Defense also got on board, looking for fuel cells that could run on diesel.
By 2014, SAFCell had delivered to both customers, and the Norwegian company had built an auxiliary power unit around the technology. It was expensive, though, and impatient investors shuttered the operation.
On the military side, SAFCell partnered with another fuel cell company, UltraCell, to successfully develop wearable propane-fueled units for the Army, as a lightweight power source for troops’ electronic devices. Chisholm’s company built the fuel cell stacks and UltraCell integrated them into the final system. It’s a partnership that continues to develop portable power systems for the military today.
As SAFCell looked for new markets, the oil and gas industry emerged as a clear possibility. Drilling sites typically use the pressure of natural gas escaping from the well to power pneumatic components such as the methanol injection pump that keeps a well from freezing. Then the gases—mostly methane, one of the most powerful greenhouse gas—are vented into the atmosphere. SAFCell saw an alternative: a fuel cell could run on the methanol that’s already on site for freeze protection and power non-emitting electrical components, eliminating natural gas emissions.
Benefits
“Methane mitigation is a huge focus in the oil and gas industry,” says Chisholm, noting that drillers often search their gas lines to stop gas leaks while at the same time intentionally venting the same greenhouse gases to drive equipment at the well site. “It’s outrageous, and they recognize that very soon that’s not going to be doable, whether regulation or public-image concerns bring it to an end.”
In Canada, where methane from oil and gas production accounts for 6 percent of the country’s greenhouse gas emissions, new regulations give producers four years to cut these emissions in half. These rules have created an estimated $3.1 billion market for retrofitting wells, of which Chisholm guesses about 10 percent will go to systems to power electric equipment. “That’s a big, juicy market to go after,” he says. “We like that combination of making money and going green while we’re doing it.”
Solar panels don’t generate nearly enough energy to do the job, and the only other standard alternative, thermoelectric generators, are only about 3–6 percent efficient. A SAFCell fuel cell generator, meanwhile, is about 30 percent efficient.
SAFCell’s “superprotonic” solid acid electrolyte increases efficiency not just by rapidly transferring protons from anode to cathode but also by operating at a high temperature. Heat accelerates chemical reactions, explains Narayan. His methanol cells are constrained by the boiling point of their water-based electrolyte, above which membranes dry out. The solid acid cells, on the other hand, can operate around 480 °F. “Under those conditions, oxidation of methanol occurs rapidly,” Narayan says, referring to the chemical reaction that results in usable electricity. The heat also lets the cells break down more complex fuels like diesel, natural gas, and propane.
Other, hotter fuel cells operate between 900 and 1,800 °F, increasing efficiency even further, but they’re complex and expensive. These are typically large, high-power stationary units.
“We’re kind of the intermediate solution to existing fuel cell technologies,” Chisholm says. “We look good for lower-power units in portable, mobile, or remote stationary applications. We have a nice combination of ruggedness, portability, and low cost.”
The company deployed its first five units to well sites in Canada in 2018 and was slated to deliver at least 13 more in 2019. The military remains a customer and is now funding the development of cells that can run on JP-8 jet fuel to power portable equipment. “Once we have a JP-8 system, it’s off to the races with them,” Chisholm says.
No matter how large the military and oil and gas markets turn out to be, SAFCell doesn’t intend to stop there. “We want to get the volume up and the price down so we can enter bigger markets, like range extenders on vehicles and residential heat and power units,” says Chisholm. “If we can get the price point down and extend the lifetime, we can compete in a lot of markets.”

SAFCell partnered with another company to develop lightweight, wearable propane fuel cells for the military, to power troops’ electronic equipment in the field. The Defense Department is now funding the development of similar cells that run on JP-8 jet fuel. Image courtesy of the U.S. Army

Instead of using the pressure from natural gas that escapes from oil wells to power pneumatic components and then venting the gas, drillers can use SAFCell’s fuel cells to power equipment, keeping methane, a powerful greenhouse gas, out of the atmosphere. The cells offer high efficiency at low cost and complexity.